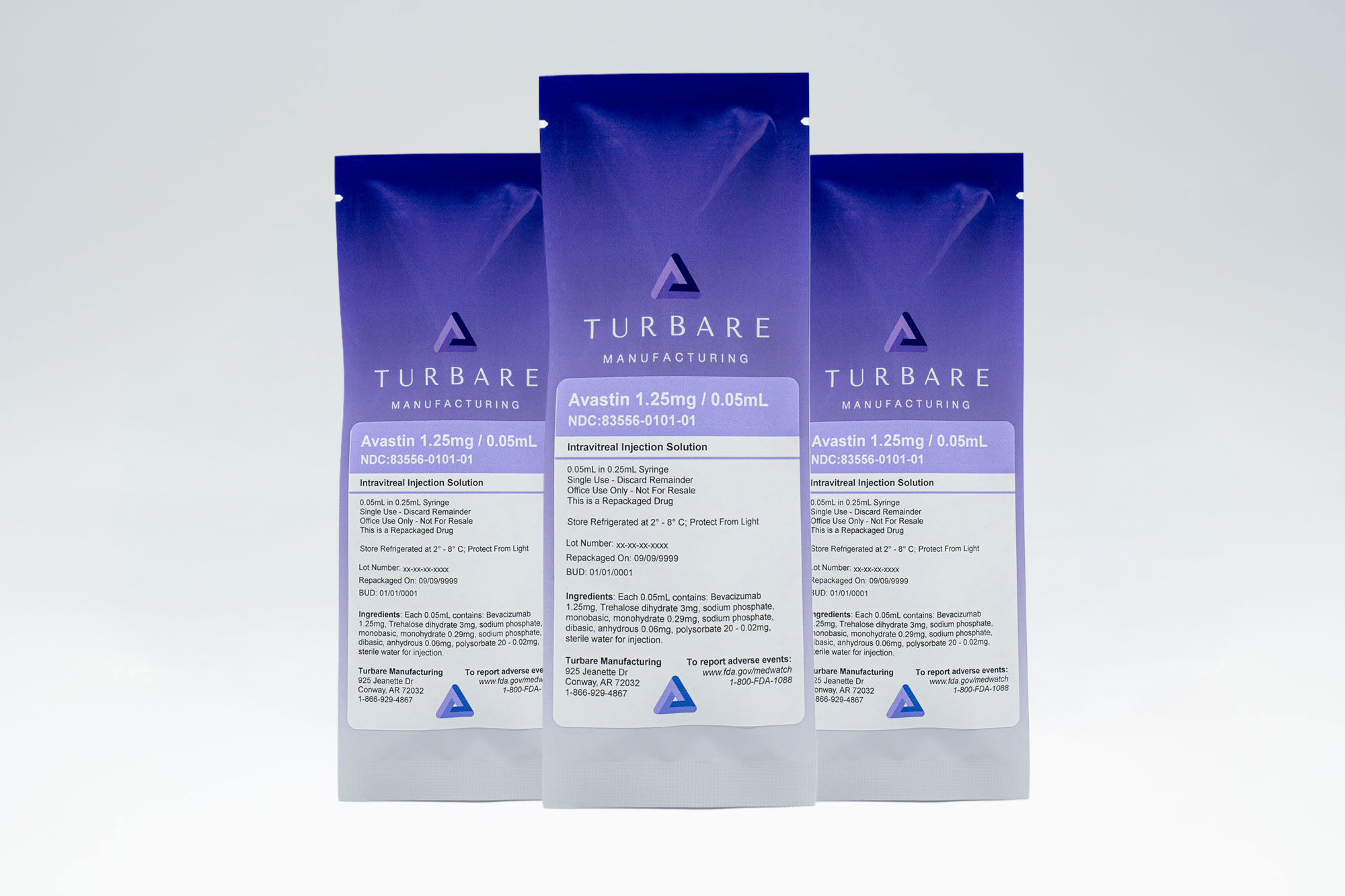
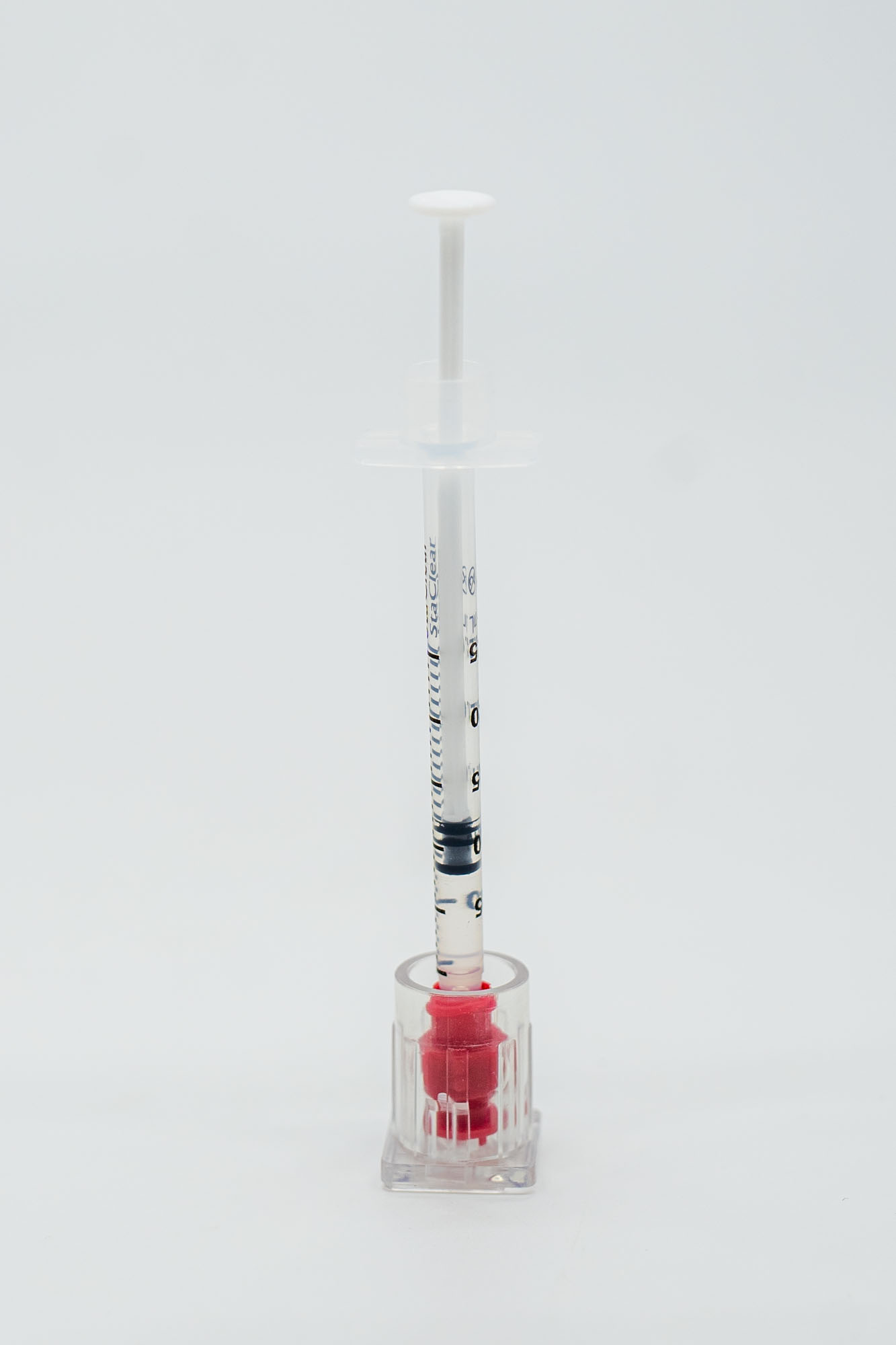
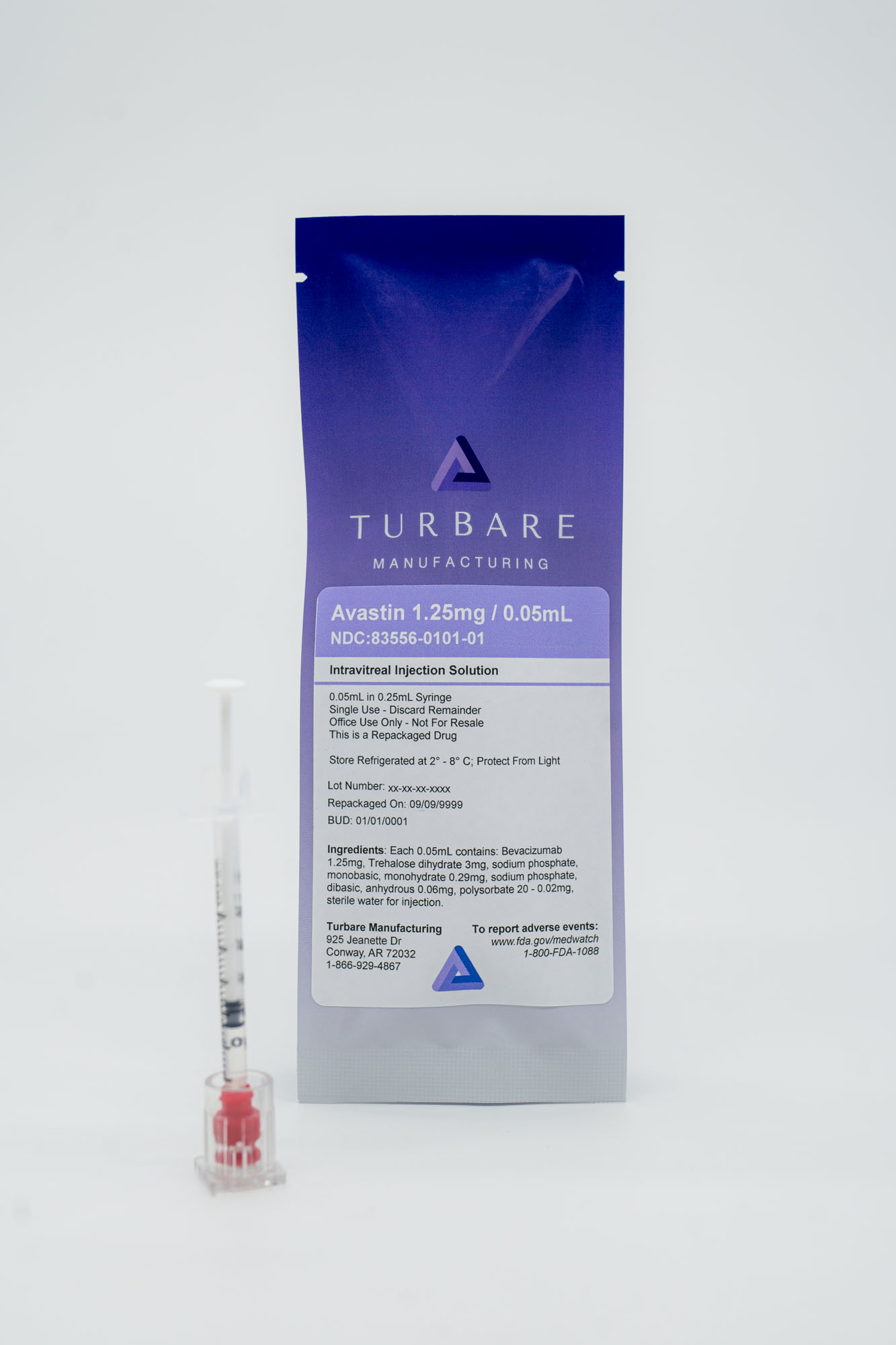
Avastin
1.25mg per 0.05mL Injection – Bevacizumab
Shorten the distance between evidence-based anti-VEGF therapy and the patients who need it. Turbare Manufacturing repackages Avastin (bevacizumab) into single-use, ready-to-administer intravitreal syringes for qualified ophthalmic providers.
For Intravitreal Use Only · Office Use Only · Single-Use Syringes
$100.00
Key Features:
- Contains Bevacizumab – a targeted anti-VEGF therapy
- Helps reduce swelling and leakage in the retina
- Used for wet AMD, DME, and RVO
- Administered via intravitreal injection by a healthcare professional
- Helps maintain and improve vision in affected patients
Made for Retinal & Comprehensive Ophthalmology Practices
Intravitreal bevacizumab has become a key anti-VEGF option in the management of neovascular retinal disease, including neovascular (wet) age-related macular degeneration and other VEGF-driven retinal vascular conditions, when used off-label at the discretion of the treating physician.
Turbare’s repackaged Avastin 1.25mg/0.05mL Injection is designed for:
Why Providers Choose Avastin from Turbare
Turbare Manufacturing is an FDA-registered, 503B specialty outsourcing facility focused on sterile, injectable medications that support critical needs in the pharmaceutical supply chain.
What sets our Avastin syringes apart:
503B Outsourcing Facility
Automated, cGMP-aligned production in a purpose-built cleanroom environment.
Focused on Sterility & Quality
Sterile preparation and packaging in a facility dedicated to injectable medications, supported by robust quality systems.

Ready-to-Administer Format
Prefilled single-use syringes at 1.25mg per 0.05mL, prepared specifically for intravitreal injection by qualified eye-care providers.
Documentation & Traceability
Lot-level documentation and labeling designed for straightforward incorporation into practice records and inventory workflows.
Quick Information
Each Turbare syringe begins with FDA-approved Avastin (bevacizumab), a recombinant humanized monoclonal antibody that binds vascular endothelial growth factor (VEGF-A) to help inhibit abnormal blood vessel growth and leakage.
Key product details:
Active Ingredient: Bevacizumab (Avastin)
Concentration: 1.25mg per 0.05mL (25mg/mL)
Route: Intravitreal injection only
Use: Office use only · Not for resale · Single-use syringe
Formulation (from source Avastin):
Bevacizumab 25mg/mL with excipients such as trehalose dihydrate, sodium phosphate, polysorbate 20, and water for injection in a preservative-free solution. For full composition and systemic indications, refer to the official Avastin prescribing information from Genentech and regulatory authorities.
Bevacizumab is a monoclonal antibody that targets VEGF-A, a key mediator of pathologic angiogenesis and vascular permeability. By binding VEGF-A, bevacizumab helps reduce abnormal blood vessel growth and fluid leakage in VEGF-driven disease processes.
In retinal care, intravitreal bevacizumab is used off-label in conditions such as neovascular age-related macular degeneration and other VEGF-mediated retinal diseases, supported by a growing body of clinical literature.
Clinical decisions, dosing, and retreatment intervals must always follow the judgment of the treating ophthalmologist based on current evidence, guidelines, and the full systemic Avastin label.
Turbare Manufacturing operates in a state-of-the-art, fully automated facility in Conway, Arkansas, designed explicitly for sterile injectable and critical medication preparations.
FDA-Registered 503B Outsourcing Facility
Registered as a 503B outsourcing facility and aligned with current good manufacturing practices (cGMP) for sterile drug production.Cleanroom Innovation
Advanced cleanroom design supports consistent environmental controls and aseptic processing.Experienced Team
Highly trained personnel with years of 503B outsourcing experience, focused on safe and reliable sterile products.Committed to the Supply Chain
Focus on sterile intravenous medications, drug-shortage preparations, and critical therapies that support care teams nationwide.
Intended Use
For intravitreal injection only.
To be administered exclusively by qualified ophthalmic professionals in appropriate clinical settings.
Before Use
Review the Avastin (bevacizumab) full prescribing information and relevant retinal disease guidelines.
Determine whether off-label intravitreal use is appropriate for the individual patient, and obtain informed consent.
Administration
Use aseptic technique in line with standard intravitreal injection protocols and your practice’s policies.
Discard any unused portion. Each syringe is intended for single use only.
(This section is informational and not a substitute for the official systemic Avastin prescribing information or applicable professional guidelines.)
This is not complete safety information; always consult the full systemic Avastin label and current retinal literature before use.
Considerations for Intravitreal Use
Intravitreal bevacizumab—while widely used—remains an off-label route for Avastin in the U.S. Published experience has described benefits alongside known ocular and systemic risks.
Serious ocular events reported with intravitreal anti-VEGF therapy, including bevacizumab, have included:
Endophthalmitis
Retinal detachment or retinal tears
Intraocular inflammation
Increases in intraocular pressure
Systemic bevacizumab carries labeled warnings for events such as gastrointestinal perforation, hemorrhage, thromboembolic events, impaired wound healing, hypertension, and renal effects, among others.
This product and webpage are intended for licensed healthcare professionals only and are not a patient information resource or advertising for direct-to-consumer use. Patients should be directed to their eye-care provider for individualized guidance.
Note: Avastin (Bevacizumab) is an off-label use for ophthalmology, originally developed for cancer treatment. However, it is widely used and recommended by specialists for retinal diseases. Always consult your doctor to determine if this treatment is right for you.
Prescription Required. For medical use only.