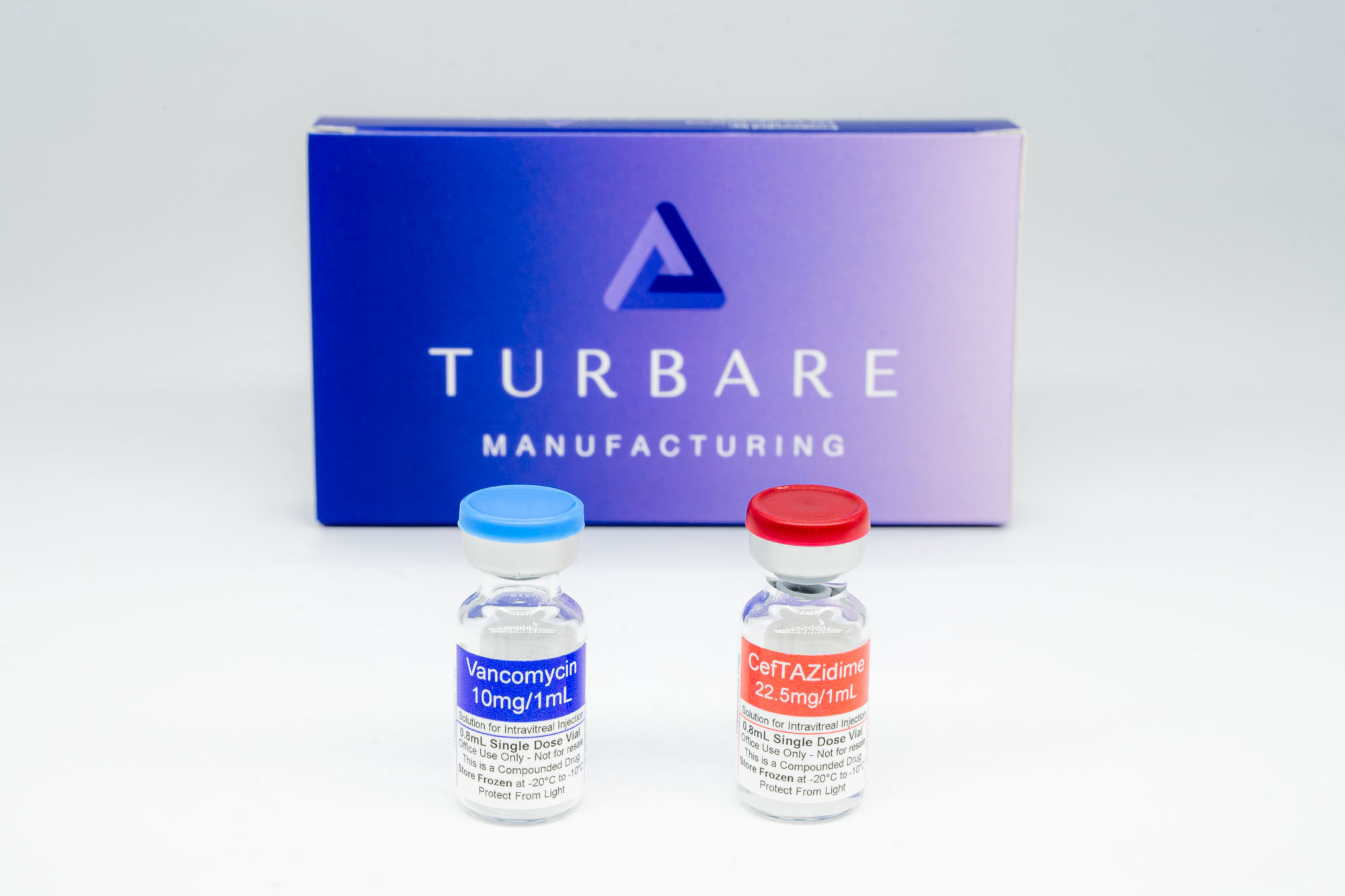
Antibiotic Kit
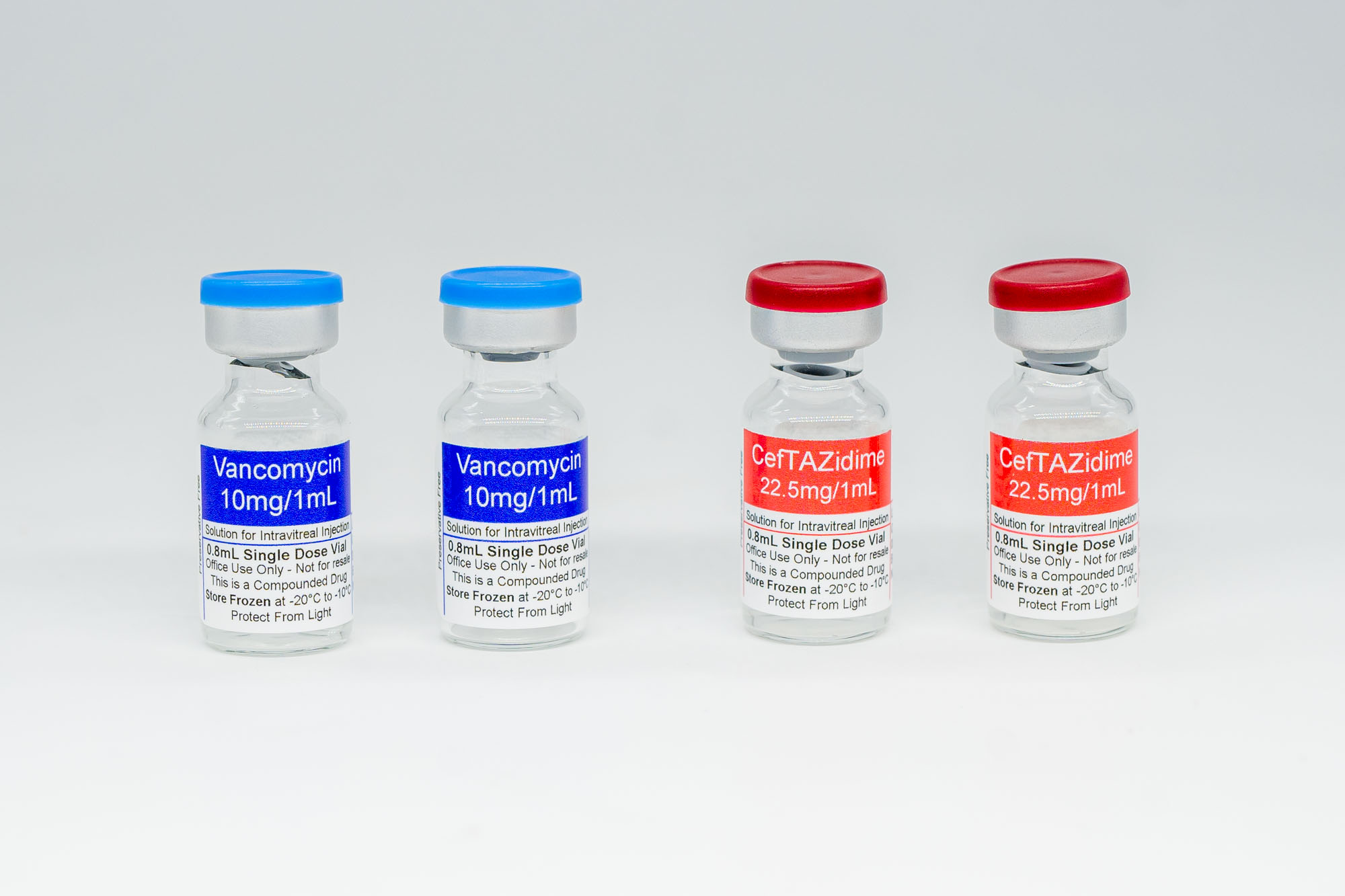
Our Antibiotic Kits are now shipping and ready to order. Each kit includes 4 vials total (2 vials of Ceftazidime + 2 vials of Vancomycin), is shipped frozen, includes free shipping, and has a 60-day expiration.
$80.00
Key Features:
- 4 vials per kit
- 60-day expiration
- Shipped frozen
- Free shipping
Each Kit Includes:
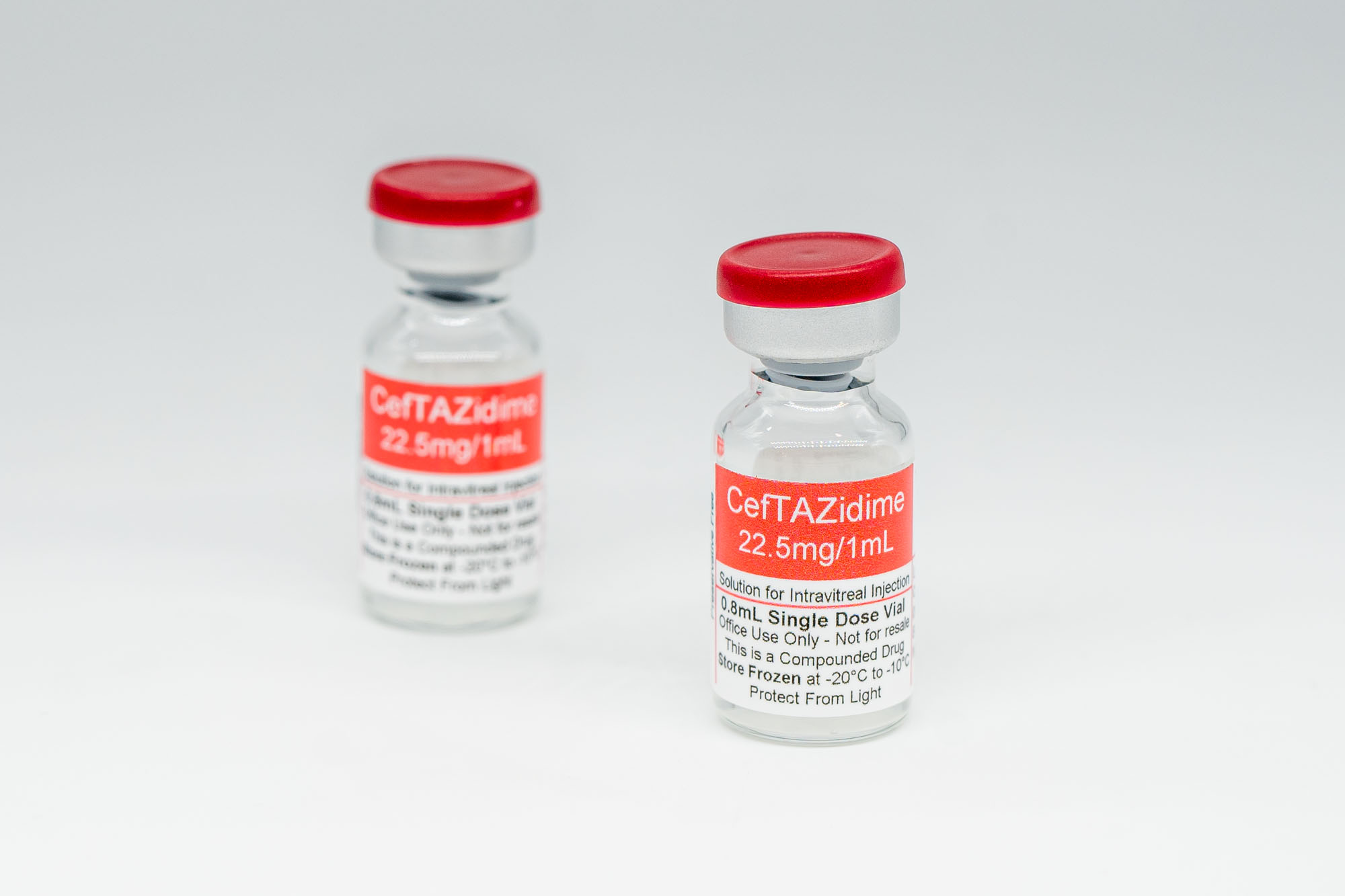
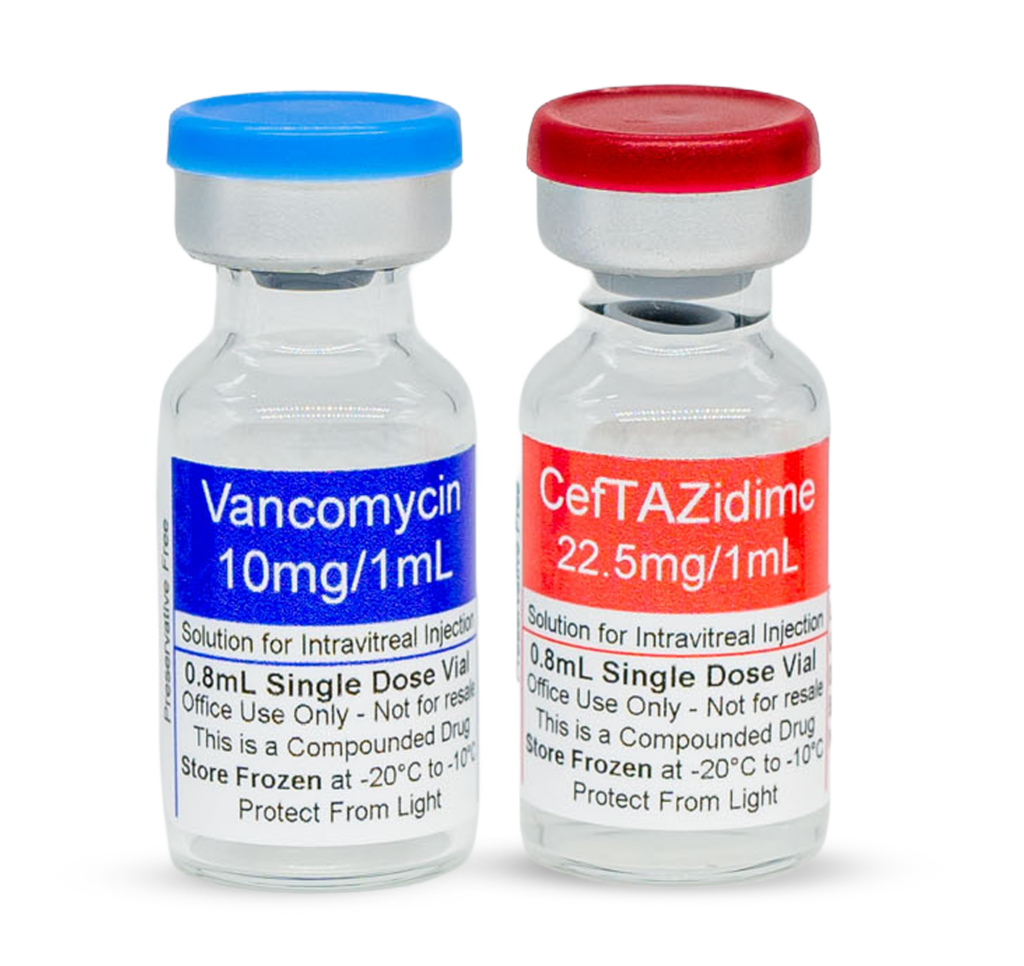
- Ceftazidime 22.5 mg/mL, 3 mL (2 vials)
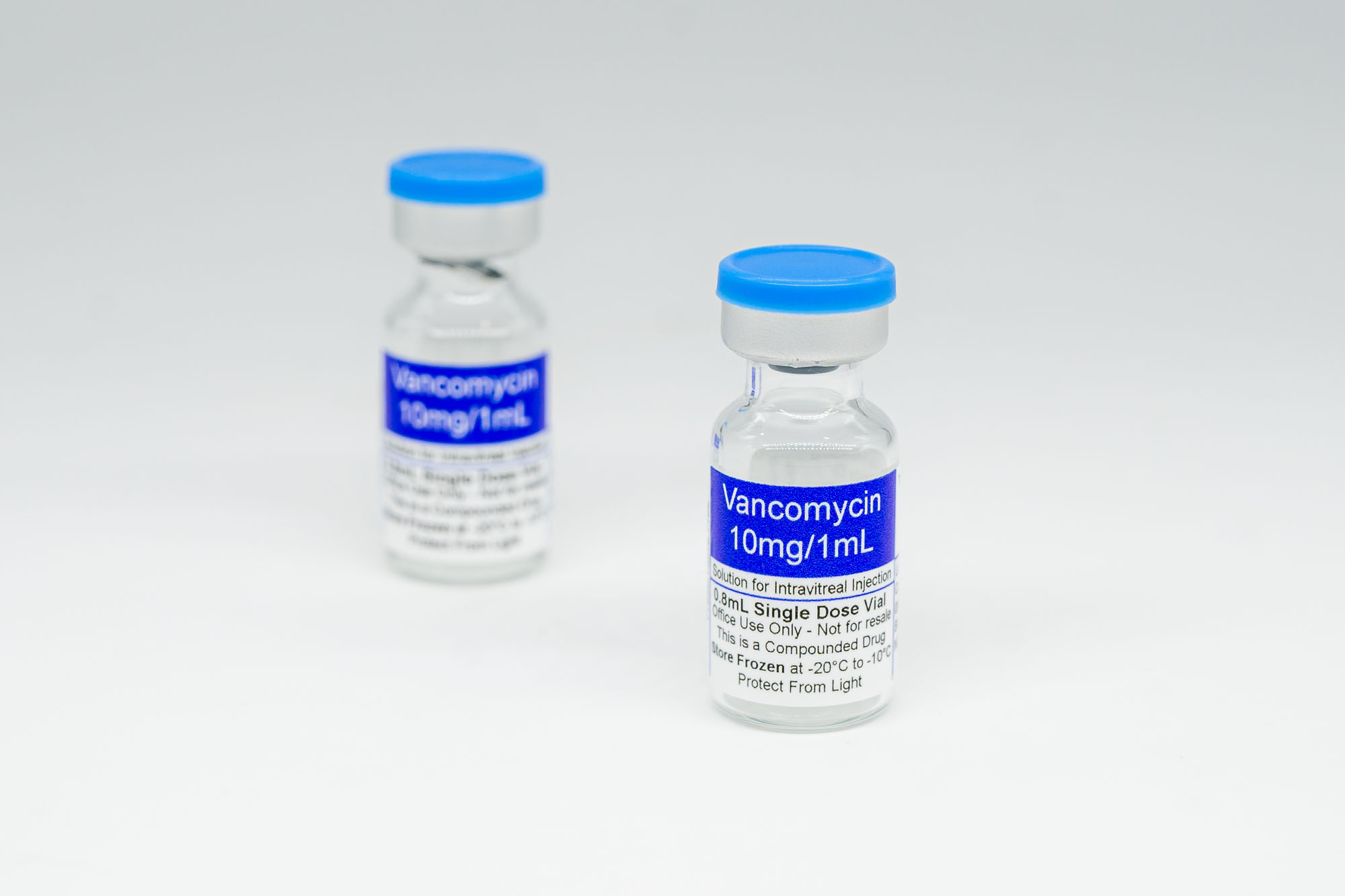
- Vancomycin 10 mg/mL, 3 mL (2 vials)
Made for Ophthalmic Practices & Care Teams
Sterile antibiotic preparations are often needed quickly and handled with strict protocol. This kit is intended for qualified clinical use by licensed providers and care teams who need consistent packaging, clear labeling, and a reliable ordering process.
Designed for:
Ophthalmic practices and ambulatory surgery centers (ASCs)
Health systems and group practices
Clinician-led teams requiring sterile, traceable vials for controlled clinical workflows
Why Providers Choose Antibiotic Kits from Turbare
Turbare Manufacturing is an FDA-registered, 503B specialty outsourcing facility focused on sterile, injectable medications that support critical needs in the pharmaceutical supply chain.
503B Outsourcing Facility
Automated, cGMP-aligned production in a purpose-built cleanroom environment.
Focused on Sterility & Quality
Sterile preparation and packaging in a facility dedicated to injectable medications, supported by robust quality systems.
Ready-to-Use Kit Format
Single-use vials packaged to simplify ordering, receiving, and clinical preparation.
Documentation & Traceability
Lot-level documentation and labeling designed for straightforward incorporation into practice records and inventory workflows.
Quick Information
Each Antibiotic Kit includes 4 vials total:
Ceftazidime 22.5 mg/mL, 3 mL (2 vials)
Vancomycin 10 mg/mL, 3 mL (2 vials)
Key product details:
Presentation: Single-use vials
Total quantity: 4 vials per kit
Expiration: 60 days
Shipping: Shipped frozen + free shipping
Ceftazidime (a cephalosporin-class antibiotic) and vancomycin (a glycopeptide-class antibiotic) are widely used antimicrobial agents. Clinical use, preparation, and administration must follow the judgment of the treating clinician, current guidelines, and your facility’s policies.
Turbare Manufacturing operates in a state-of-the-art, fully automated facility in Conway, Arkansas, designed explicitly for sterile injectable and critical medication preparations.
FDA-Registered 503B Outsourcing Facility
Registered as a 503B outsourcing facility and aligned with current good manufacturing practices (cGMP) for sterile drug production.Cleanroom Innovation
Advanced cleanroom design supports consistent environmental controls and aseptic processing.Experienced Team
Highly trained personnel with years of 503B outsourcing experience, focused on safe and reliable sterile products.Committed to the Supply Chain
Focus on sterile intravenous medications, drug-shortage preparations, and critical therapies that support care teams nationwide.
Intended use
For use by licensed healthcare professionals only
For office/clinical use only
Before use
Verify product labeling, lot information, and your facility’s handling requirements
Follow your institution’s policies and applicable professional guidelines
This is not complete safety information. Always consult applicable prescribing information for each component and follow your facility’s protocols.
Potential risks associated with injectable antibiotic use may include hypersensitivity reactions, adverse local/systemic effects, and risks related to sterile preparation and administration technique. This product page is intended for licensed healthcare professionals only and is not patient-directed information.
Prescription Required. For medical use only.
This is not complete safety information. Always consult applicable prescribing information for each component and follow your facility’s protocols.
Potential risks associated with injectable antibiotic use may include hypersensitivity reactions, adverse local/systemic effects, and risks related to sterile preparation and administration technique. This product page is intended for licensed healthcare professionals only and is not patient-directed information.
Prescription Required. For medical use only.
Use aseptic technique per your clinical protocol
Single-use vials: discard any unused portion after puncture/use
(This section is informational and does not replace institutional policy, professional guidelines, or prescribing information.)
This is not complete safety information. Always consult applicable prescribing information for each component and follow your facility’s protocols.
Potential risks associated with injectable antibiotic use may include hypersensitivity reactions, adverse local/systemic effects, and risks related to sterile preparation and administration technique. This product page is intended for licensed healthcare professionals only and is not patient-directed information.
Prescription Required. For medical use only.
Note: For professional/clinical purchasing only. Product availability may change. Please follow all applicable regulations and your facility’s handling requirements.